Millions of people rely on calcium supplements to maintain strong bones and support vital bodily functions, yet few understand what happens behind the scenes to transform raw materials into the tablets and capsules they take daily. When your doctor recommends calcium supplementation or you reach for a bottle at the pharmacy, you’re accessing products created through sophisticated manufacturing processes that convert natural calcium sources into bioavailable forms your body can actually use. Understanding how are calcium supplements made reveals why some forms work better than others and how to choose supplements that deliver maximum benefit without hidden risks.
The journey from mineral deposit to supplement bottle involves precise chemical engineering, rigorous quality control, and specialized processing techniques that directly impact how well your body absorbs and utilizes the calcium. Whether derived from ancient limestone deposits, recycled eggshells, or oyster shells, each calcium source requires specific transformation methods to become safe, effective supplement material. The manufacturing process determines not only the purity and safety of your calcium supplement but also its bioavailability—how efficiently your digestive system can extract and deliver that calcium to your bones and bloodstream.
Mining and Processing Limestone for Calcium Carbonate Supplements
The calcium carbonate in most over-the-counter supplements starts its journey deep underground in limestone quarries where ancient marine deposits have hardened over millions of years. Mining operations extract these calcium-rich deposits using controlled blasting and heavy machinery, then transport the raw material to processing facilities where it undergoes multiple refinement stages. The initial crushing reduces large rock formations to manageable chunks, followed by grinding that transforms the material into fine particles with consistent size distribution critical for pharmaceutical applications.
Why Calcination Transforms Raw Limestone into Supplement Material
The calcination process represents the critical transformation step where crushed limestone becomes reactive calcium oxide (quicklime). Heating limestone to temperatures between 900-1,000°C drives off carbon dioxide through thermal decomposition, fundamentally changing the chemical structure. This high-heat treatment requires precise temperature control—too low and the reaction remains incomplete; too high and the material sinters, reducing its reactivity for subsequent processing. Industrial kilns carefully monitor these parameters throughout the 2-4 hour calcination cycle to ensure optimal conversion without energy waste.
How Carbonation Creates Pharmaceutical-Grade Calcium Carbonate
Following calcination, manufacturers hydrate the quicklime by adding water to create calcium hydroxide slurry, then introduce carbon dioxide gas in controlled carbonation chambers. This step regenerates calcium carbonate but with dramatically different physical properties than the original limestone. The carbonation process allows precise manipulation of:
– Particle size (typically 0.5-5 microns for supplements)
– Crystal structure (calcite, aragonite, or vaterite forms)
– Surface area (higher surface area improves dissolution)
These controlled characteristics directly impact how your digestive system processes the calcium supplement. Precipitated calcium carbonate created through this method offers superior consistency and bioavailability compared to simply grinding raw limestone, explaining why it dominates the supplement market despite more complex manufacturing.
Transforming Eggshells into High-Quality Calcium Supplements
Food processing facilities generate staggering quantities of eggshell waste—approximately 94% calcium carbonate by composition—that most consumers never consider as a potential supplement source. The eggshell conversion process begins with collection from commercial kitchens and food manufacturers, followed by rigorous cleaning protocols that eliminate all organic residues. Unlike mined sources, eggshells require intensive sanitation because they originate from food production environments where pathogens could potentially contaminate the final product.
Essential Eggshell Cleaning and Sterilization Steps
Before eggshells can enter supplement production, they undergo a multi-stage purification process that typically includes:
1. Mechanical removal of membrane residues using specialized brushes
2. Soaking in food-grade sanitizers (often citric or acetic acid solutions)
3. High-temperature washing (70-85°C) to eliminate microbial contaminants
4. Final rinse with purified water to remove any residual cleaning agents
This thorough cleaning is non-negotiable—any remaining organic matter would compromise supplement safety and stability. The process typically takes 2-3 hours per batch and requires careful monitoring to ensure complete pathogen elimination while preserving the calcium carbonate structure.
Drying and Milling Techniques for Optimal Bioavailability
After cleaning, eggshells must be completely dried to prevent microbial growth during storage and processing. Manufacturers employ three primary drying methods:
– Conventional oven drying: Most economical but risks thermal degradation at high temperatures
– Freeze drying: Preserves natural structure but increases production costs significantly
– Solar drying: Sustainable option with longer processing times (24-48 hours)
The dried shells then undergo fine grinding to achieve particle sizes between 5-50 microns, with smaller particles enhancing dissolution rates in the digestive tract. Advanced facilities use planetary ball mills that create nano-structured calcium with dramatically increased surface area, potentially improving absorption by up to 30% compared to conventionally processed materials.
Manufacturing Calcium Citrate Through Acid Dissolution
Calcium citrate supplements follow a fundamentally different production path than calcium carbonate, starting with a controlled chemical reaction between calcium sources and citric acid. This process begins with either calcium carbonate or calcium hydroxide reacting with food-grade citric acid under precisely monitored conditions. The reaction stoichiometry must maintain a 3:1 molar ratio of citric acid to calcium to ensure complete conversion to calcium citrate tetrahydrate—the form used in supplements.
Why Temperature Control Determines Final Product Quality
The acid dissolution reaction generates significant heat that must be carefully managed to prevent decomposition of the citric acid or formation of unwanted byproducts. Manufacturers maintain reaction temperatures between 60-80°C using jacketed reactors with cooling capabilities. Exceeding 90°C risks caramelizing the citric acid, while temperatures below 50°C slow the reaction to commercially unviable rates. The entire dissolution process typically takes 1-2 hours with continuous pH monitoring to confirm reaction completion at approximately pH 6.5-7.0.
Crystallization and Drying Methods for Maximum Stability
After complete dissolution, manufacturers cool the solution to induce crystallization of calcium citrate tetrahydrate. The cooling rate dramatically affects crystal size and morphology—rapid cooling creates small, uniform crystals ideal for tablet compression, while slow cooling produces larger crystals better suited for encapsulation. The crystallized product then undergoes centrifugation to separate crystals from mother liquor, followed by drying at 60-70°C to achieve the target moisture content of less than 5%. This drying step requires 4-6 hours and must avoid excessive heat that could dehydrate the tetrahydrate structure, compromising stability.
Avoiding Heavy Metal Contamination in Calcium Production
All calcium supplement manufacturers face the critical challenge of preventing heavy metal contamination, particularly lead, arsenic, cadmium, and mercury. Natural calcium sources like limestone, dolomite, and oyster shells often contain trace amounts of these elements absorbed from their environments over geological time scales. The FDA mandates maximum lead levels of 10 parts per million (ppm) in dietary supplements, requiring manufacturers to implement rigorous testing protocols at multiple production stages.
Three-Stage Testing Protocol for Supplement Safety
Reputable manufacturers employ a comprehensive contamination control system that includes:
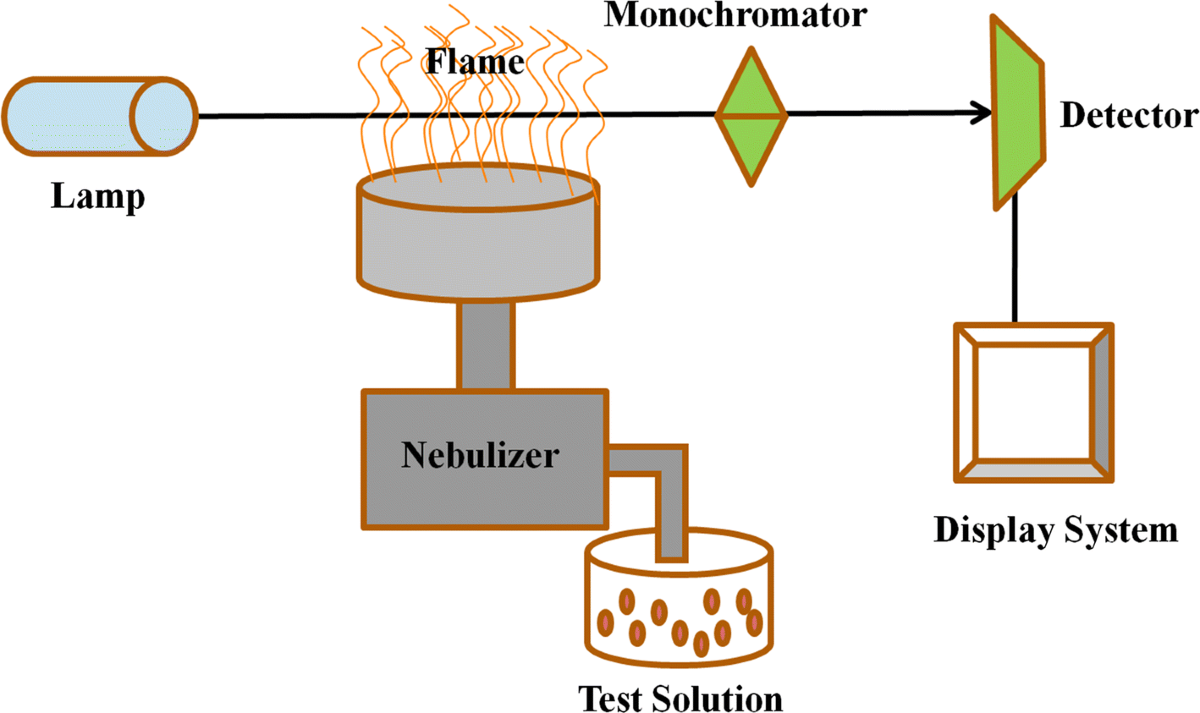
1. Incoming raw material testing: Every shipment of limestone, eggshells, or other source materials undergoes atomic absorption spectroscopy to detect heavy metals
2. In-process verification: Samples taken during manufacturing confirm processing steps haven’t introduced contaminants
3. Finished product certification: Each production batch receives full heavy metal analysis before release
This multi-stage approach catches potential contamination early, preventing costly batch rejections at final stages. The entire testing process adds approximately 24-48 hours to production timelines but is essential for consumer safety.
Why Eggshell Sources Offer Lower Contamination Risk
Eggshells present a significant advantage over mined sources regarding heavy metal contamination. Since chickens metabolize and excrete heavy metals before shell formation, eggshell calcium typically contains lead levels 5-10 times lower than limestone-derived products. Research shows average lead content in eggshell calcium measures just 0.2-0.5 ppm compared to 2-5 ppm in commercial limestone products. This natural purification process makes eggshell-derived supplements particularly valuable for vulnerable populations like children and pregnant women.
Comparing Supplement Forms: Manufacturing Impacts on Absorption
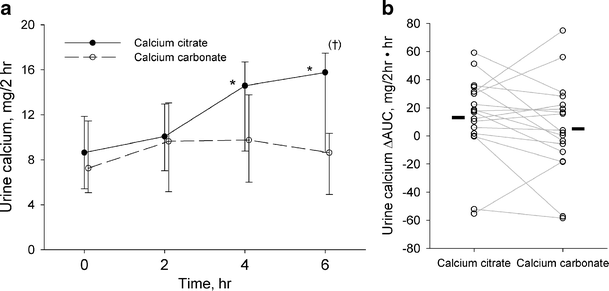
The manufacturing process directly determines how efficiently your body absorbs calcium from supplements, with significant differences between forms. Calcium carbonate requires stomach acid for optimal dissolution, making it less effective when taken without food. The manufacturing process for carbonate supplements often includes buffering agents to modify dissolution rates, but they still deliver only 27-39% absorption under ideal conditions. In contrast, calcium citrate’s manufacturing process creates a pre-dissolved form that achieves 20-25% absorption regardless of stomach acidity.
Why Particle Size Matters for Calcium Bioavailability
Manufacturers carefully control particle size during processing because smaller particles dissolve faster in the digestive tract. Research shows calcium carbonate particles below 10 microns achieve up to 40% higher absorption than larger particles. Advanced milling techniques can produce particles as small as 0.5 microns, but this increases production costs significantly. Most commercial supplements maintain particle sizes between 5-20 microns as the optimal balance of bioavailability and manufacturing feasibility.
How Chelation Technology Enhances Absorption
Premium calcium supplements use chelation technology during manufacturing to bind calcium ions to amino acids like glycine. This process creates stable complexes that resist precipitation in the digestive tract and may utilize specialized amino acid transport mechanisms. The chelation reaction requires precise pH control (typically 6.5-7.5) and temperature (40-50°C) over 2-4 hours to ensure complete complex formation. While more expensive to produce, these chelated forms achieve absorption rates up to 50% higher than standard calcium carbonate, particularly valuable for individuals with compromised digestive function.
Sustainable Manufacturing Innovations Transforming the Industry
The calcium supplement industry is rapidly adopting waste-to-resource manufacturing models that convert materials destined for landfills into valuable health products. Eggshell recycling represents the most promising sustainable approach, with food processing facilities generating approximately 150,000 tons of eggshell waste annually in the United States alone. Converting this waste stream into calcium supplements addresses both environmental concerns and public health needs while reducing production costs by up to 30% compared to mined sources.
Three Breakthrough Technologies Reducing Environmental Impact
Innovative manufacturers are implementing advanced processing methods that minimize environmental footprints:
– Supercritical CO2 extraction: Uses carbon dioxide under high pressure to extract calcium without chemical solvents
– Enzymatic processing: Employes food-safe enzymes to remove organic matter from eggshells, reducing water and energy use
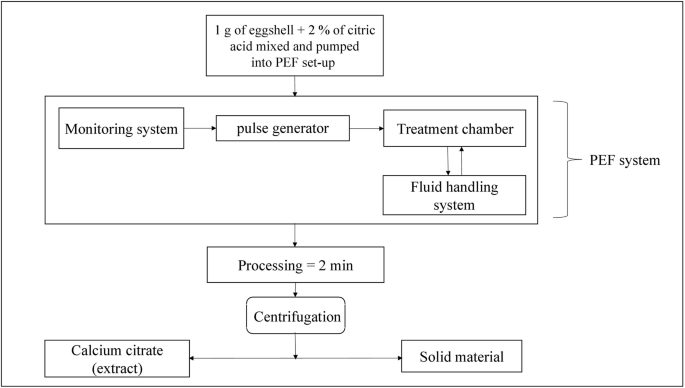
– Pulsed electric field technology: Creates microscopic channels in eggshell structure, enhancing calcium release with minimal energy input
These technologies reduce water consumption by up to 70% and energy use by 40-60% compared to traditional manufacturing methods, while producing calcium with superior bioavailability characteristics.
Why Sustainable Manufacturing Means Better Supplements
Contrary to assumptions that eco-friendly processes compromise quality, sustainable calcium supplement production often yields superior products. Eggshell-derived calcium demonstrates 15-20% higher bioavailability than limestone-based products due to its natural nano-structured morphology. The manufacturing process for recycled eggshell calcium avoids the high-temperature calcination required for mined sources, preserving natural crystalline structures that enhance dissolution in the digestive tract. As consumer demand grows for environmentally responsible products, these sustainable manufacturing innovations are becoming industry standards rather than niche alternatives.
Choosing the Right Calcium Supplement Based on Manufacturing Quality
When selecting a calcium supplement, understanding how are calcium supplements made provides crucial insight beyond basic ingredient lists. Look for products that specify their source material (e.g., “eggshell-derived” or “pharmaceutical-grade limestone”) and manufacturing processes. Reputable brands will disclose third-party testing results for heavy metals and provide transparency about their production methods.
Critical quality indicators to verify:
– Certificate of Analysis showing heavy metal testing
– Clear specification of elemental calcium content
– Manufacturing location with GMP certification
– Expiration date with stability testing data
Avoid supplements that list vague ingredients like “calcium complex” or “proprietary blend” without disclosing specific forms and quantities. The most transparent manufacturers proudly share details about their sustainable sourcing and advanced processing techniques because they understand that how are calcium supplements made directly impacts how well they work for you.
The calcium supplement industry continues evolving toward more sustainable, transparent, and scientifically advanced manufacturing practices that deliver better products while reducing environmental impact. By understanding the journey from raw material to supplement bottle, you can make informed choices that support both your health and the planet. Whether you choose traditional calcium carbonate, innovative eggshell-derived products, or specialized chelated forms, the manufacturing process ultimately determines whether that calcium reaches your bones where it’s needed most.